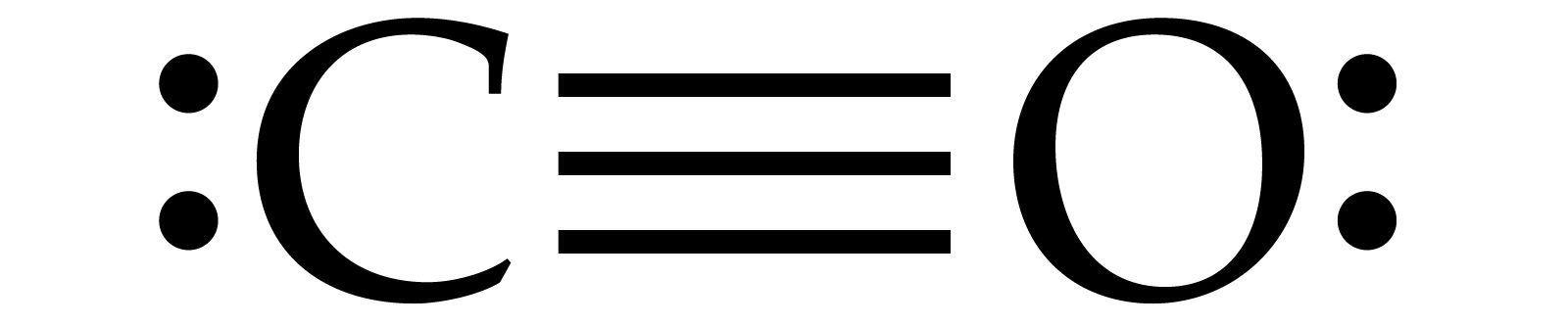
Usually, the sulfur recovery level is set by the allowable emissions of sulfur from the tail-gas incinerator. The reduction of carbonyl sulfide, carbon disulfide, sulfur dioxide, and sulfur vapor in Claus tail gas to hydrogen sulfide is necessary when sulfur recovery of 99.9+% is required. Speight Ph.D., D.Sc., in Natural Gas, 2007 7.6.5 Hydrogenation and Hydrolysis Processes Resin synthesis, pharmaceuticals, agriculture, mining, rubber Consult a physicianĬauses damage to organs through prolonged or repeated exposure if inhaled If not breathing, give artificial respiration. If breathed in, move person into fresh air. Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician Maximum exposure concentration NIOSH-IDLHĪnimal testing, acute toxicity, Rat oral LD50Īnimal testing, acute toxicity, Mouse oral LD50Īnimal testing, acute toxicity, Rat inhalation, LC50
CARBON DISULFIDE FORMULA FULL
The only respirators recommended for fire fighting are self-contained breathing apparatuses that have full facepieces and are operated in a pressure-demand or other positive-pressure mode. If employees are expected to fight fires, they must be trained and equipped in OSHA 1910.156.

If cooling streams are ineffective (venting sound increases in volume and pitch, tank discolors, or shows any signs of deforming), withdraw immediately to a secure position. From a secure, explosion-proof location, use water spray to cool exposed containers. Notify local health and fire officials and pollution control agencies. If material or contaminated runoff enters waterways, notify downstream users of potentially contaminated waters. Use dry chemical, carbon dioxide water spray or alcohol foam extinguishers. Hazardous decomposition includes sulfur dioxide, carbon disulfide and oxides of nitrogen and carbon. Pohanish, in Sittig's Handbook of Pesticides and Agricultural Chemicals (Second Edition), 2015 Fire Extinguishing The rate constant for CS 2 + O 2 (S22, S23), important for initiation, and the CS 2 + O branching fraction, which is controlling the chain-branching rate, are both uncertain at elevated temperatures and more work is needed to resolve the discrepancies. This indicates that the balance between initiation and chain branching in the model at higher temperatures is not correct. At higher temperatures, such as in the shock tube experiments, the model tends to overpredict both the induction time for CS 2 and the formation rate of atomic O upon ignition. conclude that the modeling predictions are most accurate at low temperatures where calculated ignition delays and explosion limits are in good agreement with observed values. observed a similar deviation when comparing their predictions to the flow reactor results of Homann et al. However, at 1615 K both the induction time for CS 2 and the formation rate of O atoms upon ignition are overpredicted. 15 shows that the O-atom profiles are predicted well at 18 K. The symbols mark experimental data while solid lines denote model predictions. The experimental data are taken from Saito et al.

Comparison of experimental and predicted mole fractions of atomic oxygen for oxidation of CS 2 in a shock tube.
